Antibiotics
The Antibiotics we offer at Plant Cell Technology have different liquid Penicillin-Streptomycin Solutions.
Moreover, these solutions may contain: Penicillin, Streptomycin, Glutamine, L-glutamine , and Saline Sterile-Filtered solution.
The concentration of these solutions can be from 50x to 100x and the sizes are 100ML, 6x100ML.
Penicillin-Streptomycin is a dual antibiotic solution we often use it as a supplement to cell culture media to control bacterial contamination.
Therefore, The Penicillin-Streptomycin Solution is quite effective against most gram positive and gram negative bacteria.
We produce and test all of our antibiotics in accordance with all relevant standard operating procedures.
In conclusion, all antibiotic products meet or exceed all Caisson Laboratories quality standards.
Showing all 5 results
-
 PSL03
PSL03Penicillin-Steptomycin Solution 50X.
Penicillin-Steptomycin Solution 50X.
-
 PSL01
PSL01Penicillin-Streptomycin Solution
Contains 10,000 U/ml of penicillin and 10,000 µg/ml streptomycin in a 0.9% saline sterile-filtered solution.
**Perishable Product Only ships Overnight Mondays-Wednesdays, no major holidays**
**PREORDER NOW: Lead Time December 2024
-
 PSL02
PSL02Penicillin-Streptomycin Solution (Modification 2)
Contains 10,000 U/ml of penicillin, 10,000 µg/ml streptomycin and 29.2 mg/ml L-glutamine in a 0.9% saline sterile-filtered solution.
-
 PSL04
PSL04Stabilizied Glutamine-Penicilin-Streptomycin Solution, 100X
Stabilizied Glutamine-Penicilin-Streptomycin Solution, 100X
-
 T034
T034Timentin
15:1 Ticarcillin Disodium Salt / Potassium Clavulanate Mixture
Verification of Antibiotic Potency
Potency is a measure of activity that generally describes the efficacy of an antibiotic. This measurement is necessary because purity is not a strict indicator of an antibiotics effectiveness on microorganisms.
Caisson Laboratories verifies potency values by a method derived from United States Pharmacopeia <81>, specifically the Cylinder-Plate Assay. An antibiotic of known potency is used in various concentrations to form a standard curve plotted with the concentration (x-axis) and zone of inhibition size (y-axis). The antibiotic that is to be verified is compared against that standard curve to determine its potency. It is desirable that more than one antibiotic of known concentration (the controls) are used. 3 replicates for each control are used to account for variability in microbial assays.
Caisson Laboratories follows the outline below for Verification of Antibiotic Potency:
1. Preparation of dilutions for control antibiotics and testing antibiotic
The control is diluted to 5 concentration standards that will later (in combination with measured zone of inhibition size) make up the standard curve used to verify potency. The unknown testing antibiotic is diluted to a concentration assumed to be comparable with the median concentration of the control.
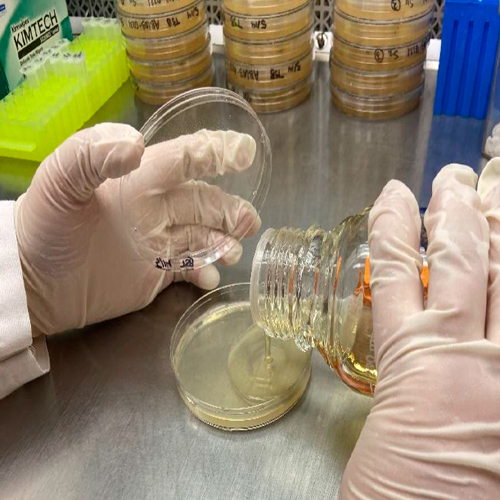
2. Preparation of sterile nutrient agar that will be inoculated with a microorganism that the antibiotic is effective against
Nutrient agar is prepared and autoclaved to ensure sterility and allowed to cool. Nutrient agar that is favorable for the inoculation organism is used.
3. Inoculation and pouring of nutrient agar into labeled plates
Stock culture will be dispensed into the nutrient agar and dispersed evenly throughout, at which point the agar will be poured onto plates that are labeled with concentration standard. Depending on the microorganism being used, the inoculated agar will be poured onto either an empty plate or a base layer of agar. Agar will be allowed to cool.
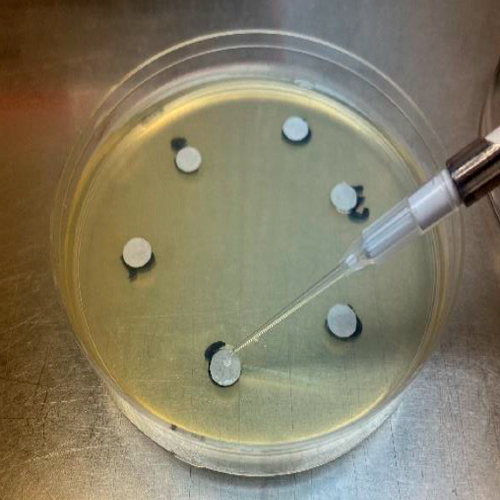
4. Placing of sterile disks on the 6 locations of the agar plate
Sterile disks are placed onto each plate with flamed forceps. These will be the locations of the dilutions of antibiotic.
5. Dispensing of control and testing dilutions onto disks
Dilutions of control and testing antibiotic are dispensed onto the discs in the specified locations. Standard 3 dilutions are present on each plate to account for variability.
6. Incubation of plates
Plates are incubated at the appropriate temperature for the amount of time it takes to develop distinguishable zones of inhibition.
7. Measuring of zones of inhibition
Zones are measured using an electronic caliper.
8. Statistical analyses of zone sizes and verification of potency
 Zone diameters are recorded into a spreadsheet that calculates averages and corrected averages. Regression statistics are run and calculations are performed to verify potency values within a specified range.
Zone diameters are recorded into a spreadsheet that calculates averages and corrected averages. Regression statistics are run and calculations are performed to verify potency values within a specified range.